This newsletter was sent out on Thurs, Jul 26; sign up to receive the next one at www.openprescribing.net
We do the stats to make your life easy…
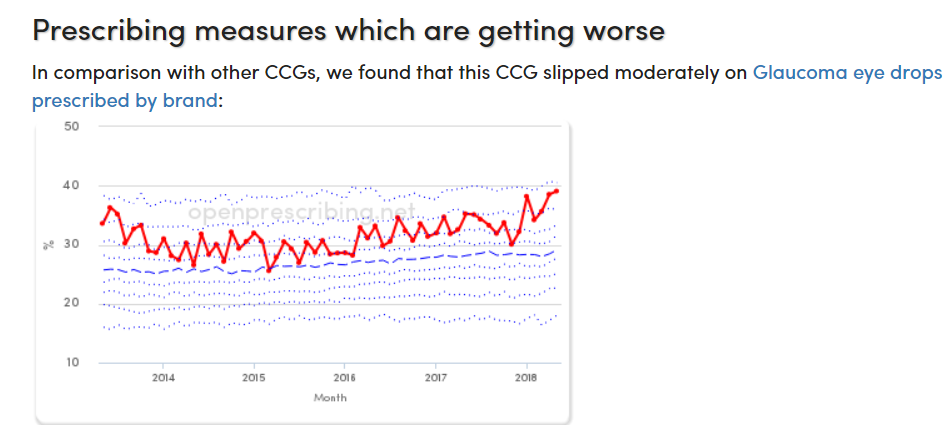
At OpenPrescribing we hide complicated data science “under the bonnet”: we want data to be easy, to come alive and tap you on the shoulder when there’s a problem. As you know we have an “alerts” service, open and free to anyone, that emails you if there’s a new issue with the prescribing at any practice. But how does it all work? Making a tool that sends an alert when a practice hits the highest 10% in the country for a given prescribing measure was easy. But we wanted to spot problems and send alerts much sooner than that: when a practice begins to drift in comparison to its peers over time, well before it becomes an extreme outlier. Our most recent paper shows the inner workings of this service. If you’re really geeky, you can read the paper here. Or you can just sign up and use the service!
New measures categories
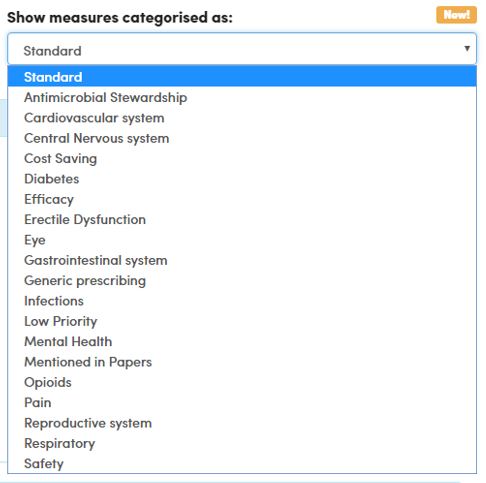
A long-awaited new feature has been implemented on our CCG and practice dashboards: you can now filter measures by category to view a small group of related measures together. You can also browse the categories here for all CCGs, and read more on our blog.
These tags will evolve as new measures are created and we would welcome any feedback and suggestions.
OpenPrescribing comes to you

This is Brian, one of our pharmacists, presenting at an event in Worcester for a large group of pharmacists working in general practices. Would you like to hear Brian talk about prescribing variation and how to use openprescribing.net to quickly audit prescribing behaviour in your area?
If you are running an event for doctors, pharmacists, nurses or anyone interested in using data to improve care, please contact feedback@openprescribing.net. Brian will try to get your event over the coming months.
Prescribing data update!
We’ve updated OpenPrescribing with May’s data. Head over to www.openprescribing.net to see more.
This is the second month of data including the new merged CCGs - we’ve put a lot of work into making these CCGs appear consistently over the 5 years of data we show, but please let us know if you spot anything that doesn’t look right.
In other news…
Unreported Trial of the Week
Following the launch of our FDAAA TrialsTracker, we have begun writing a series for the BMJ, “Unreported Trial of the Week”.
Each week we profile a new unreported clinical trial that has not reported results in accordance with their legal requirements. The last two instalments cover a statin therapy for acute recovery in stroke (NCT01976936) that has not resubmitted results during the ClinicalTrials.gov quality control process and a trial of pharmacological options to aid in fear eradication therapy (NCT00674570).
Sign up
You can sign up to receive the newsletter on www.openprescribing.net


